

 2025.12.26
2025.12.26Tired of pitfalls in synthesizing CAR mRNA yourself?
Struggling with tLNP antibody conjugation?
Waiting for materials slowing down your experiments?
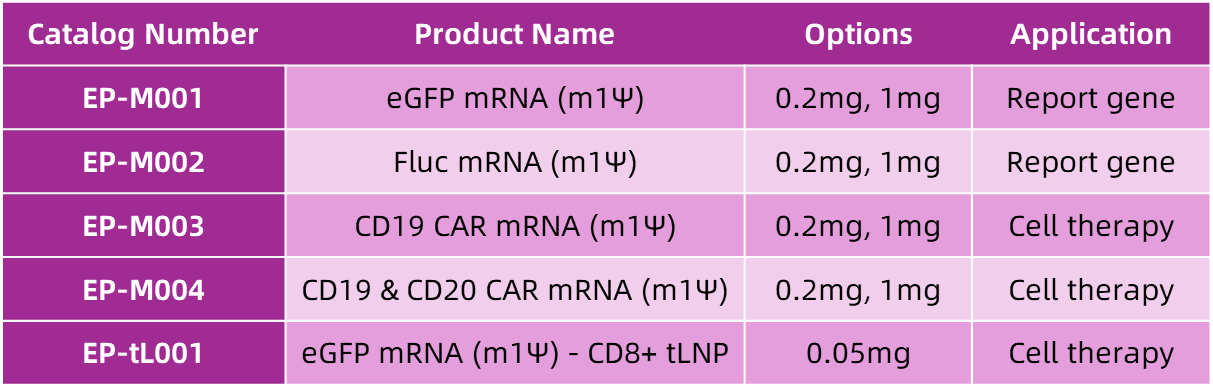
To help researchers more conveniently access validated mRNA and mRNA-tLNP, Enorna officially launches its first batch of ready-to-use mRNA & tLNP off-the-shelf products, providing customers with standardized, high-quality solutions.
The initial product lineup includes reporter gene mRNA, CAR mRNA for cell therapy, and targeted lipid nanoparticles (tLNP), precisely matching different research directions.
Highlights of the Product Batch:
Manufactured using industrial-grade preparation and purification processes and subjected to stringent quality control (QC) protocols to ensure each batch meets high-quality standards.
Case Study 1: High-Level Expression of eGFP mRNA in Cells
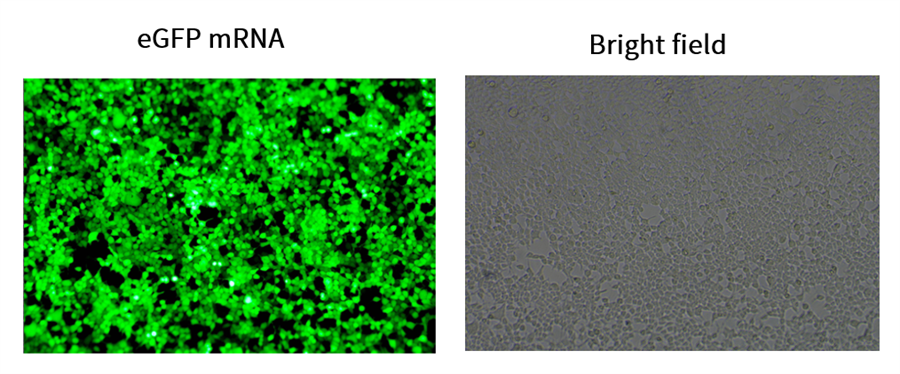
To validate the expression efficiency of Enorna's off-the-shelf mRNA products, we delivered our eGFP mRNA (m1Ψ) into 293T cells via transient transfection. Results showed that over 95% of cells were eGFP-positive 24 hours post-transfection. Our off-the-shelf products provide researchers with a high-quality, ready-to-use standard experimental control, ensuring reliable and reproducible results.
Expression of Enorna eGFP mRNA in 293T cells at 24h
Case Study 2: CD8 Antibody Conjugation Efficiency of eGFP mRNA CD8+ tLNP Reaches ~90%
Using nanoflow cytometry to detect the positive rate of CD8 antibodies on the surface of eGFP mRNA CD8+ tLNP nanoparticles, we currently achieve approximately 90% antibody-positive rate. This ensures good CD8 targeting and delivery efficiency, providing researchers conducting in vivo CAR/targeted delivery studies with a high-quality standard experimental control to ensure reliability and smooth progress in their research.
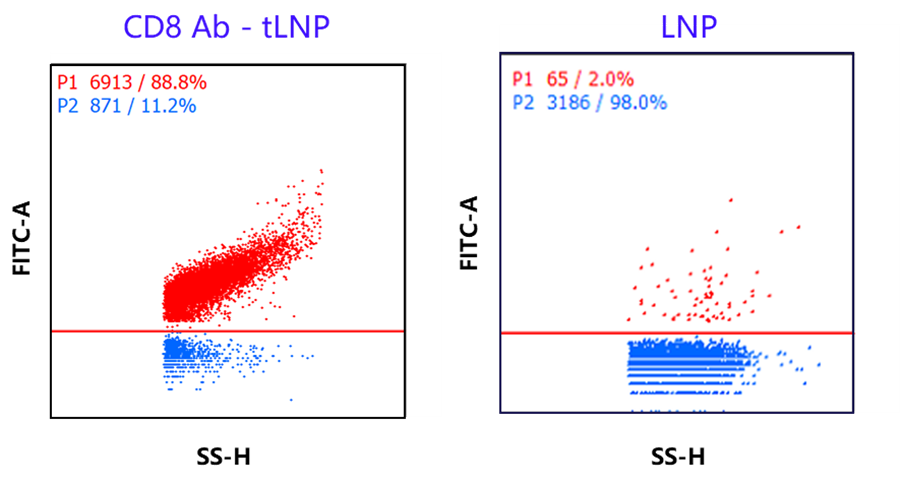
Detection of CD8 Antibody Positive Rate for eGFP mRNA CD8+ tLNP via Nanoflow Cytometry
Reach Out Today
Contact us
EPI-003, has received Investigational New Drug (IND) clearance from the U.S. Food and Drug Administration (FDA) for the treatment of chronic hepatitis B (CHB).

To help researchers more conveniently access validated mRNA and mRNA-tLNP, Enorna officially launches its first batch of ready-to-use mRNA & tLNP off-the-shelf products, providing customers with standardized, high-quality solutions.