

Lipid nanoparticles (LNPs) provide essential protection for nucleic acids and enable efficient, targeted delivery in vivo. Their stability, targeting precision, and safety profile are critical to therapeutic efficacy and clinical success.
Our integrated LNP platform offers end-to-end development and GMP manufacturing—from novel lipid screening and formulation design to process optimization and commercial-scale production. We deliver robust, scalable LNP systems to advance innovative therapies in areas such as in vivo CAR‑T, gene editing, and mRNA vaccines, accelerating their transition from development to the clinic.


IIT Batch Production & Supply

LNP Formulation Development
Analytical Development
Regulatory Support

GMP Production for Clinical Supply


LNP Encapsulation


TFF


Fill/finish


Release Test
The development of various novel lipids is essential to achieve targeted delivery beyond the liver.
We offer a diverse library of lipids with different targeting moieties for screening.
We have experience in developing and scaling up more than 25 novel lipid formulations.
Leveraging advanced microfluidic technology, we achieve efficient and stable mRNA-LNP encapsulation, ensuring an encapsulation efficiency (EE%) > 95%, uniform particle size distribution (PDI < 0.2), and high batch-to-batch consistency.
Maximum production speed 9 L/h (150 mL/min) with less than 20 mL of waste per run, supporting continuous production and highly reproducible batches.
High-concentration mRNA-LNP encapsulation technology, optimized for applications such as gene therapy and protein replacement therapy.
Successfully delivered GMP-grade LNP drug products at 50 L scale.
Proprietary LNP lyophilization process significantly enhance long-term stability of drug products.
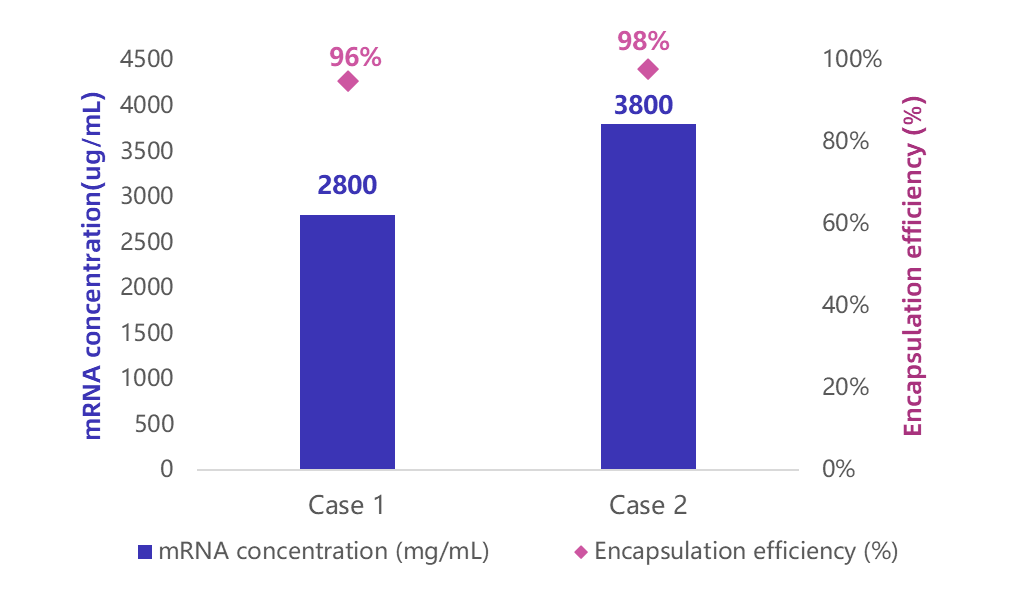
For high-dose mRNA applications, traditional low-concentration formulations are limited by large volumes and repeated injections. Enorna high-concentration LNP encapsulation enables potent, small-volume delivery.
Our optimized process delivers stable formulations with mRNA concentrations up to 3.8 mg/mL and >95% encapsulation efficiency, enhancing both druggability and clinical potential.
Enorna’s analytical platform supports the full release testing and characterization of LNP formulations, covering all Critical Quality Attributes (CQAs) required for quality assessment and regulatory submission. Key release parameters include fill volume, sub-visible and visible particles, concentra
Filling Volume: Extraction volume method
Sub-visible Particles: Microscopic counting method
Particle Size & Zeta Potential: Nano-particle size and zeta potential analysis
Encapsulation Efficiency (EE%): RiboGreen
Concentration: RiboGreen
Reach Out Today
Contact us