

With years of focused experience in nucleic acid therapeutics, Enorna provides end‑to‑end RNA drug substance development, scale‑up, and GMP production. Our platform supports linear mRNA, circRNA, and saRNA, and has been deployed across diverse modalities including in vivo CAR‑T, gene editing, personalized cancer vaccines, and protein replacement therapies. We deliver high‑integrity, low‑immunogenicity RNA to accelerate your candidate from development to clinical supply.
Enorna provides mRNA drug substance production solutions across all development stages, fully supporting needs from exploratory research to commercial-scale production.


In vitro Transcription (IVT)


Chromatographic Purification


Ultrafiltration


Release Test
In vitro transcription (IVT) is a process of efficiently synthesizing RNA from high-purity plasmid templates through a series of enzymatic reactions.
Broad RNA Synthesis Expertise – Extensive experience in producing linear mRNA, circular RNA (circRNA), and self-amplifying RNA (saRNA).
High Efficiency Capping - Co-transcriptional or enzymatic capping with efficiency consistently >98%.
Cost‑Effective & Licensable IP - Use of patented enzymes and cap analogs under commercial licenses, reducing production costs while ensuring freedom to operate.
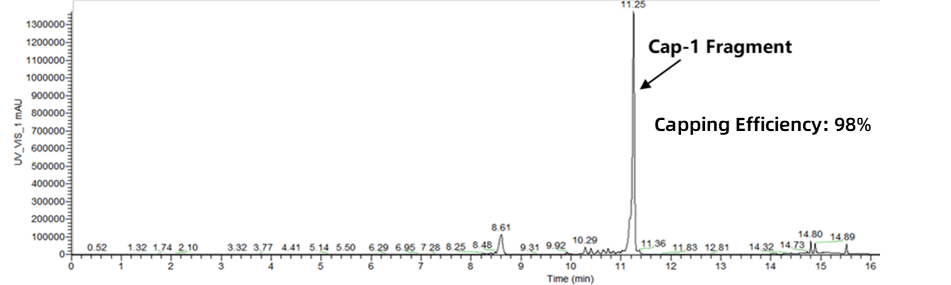
Capping efficiency directly impacts mRNA stability and translational efficiency. A high capping rate not only enhances protein expression levels but also effectively evades immune surveillance, reduces immunogenicity, improves mRNA stability, and prolongs in vivo activity duration.
Enorna stably achieves ≥98% capping efficiency, providing a foundation for mRNA stability and high-efficiency translation.
Our downstream purification process employs chromatography and tangential flow filtration (TFF) to robustly remove impurities, delivering drug substance of high purity and defined quality.
Flexible Purification Strategies – We combine multiple chromatography modes with TFF to develop tailored purification protocols optimized for specific product profiles and applications.
Effective Impurity Removal – Our process efficiently removes critical residuals including dsRNA, host DNA, uncapped/truncated RNA, and enzymes, thereby reducing immunogenicity and maximizing translational activity.
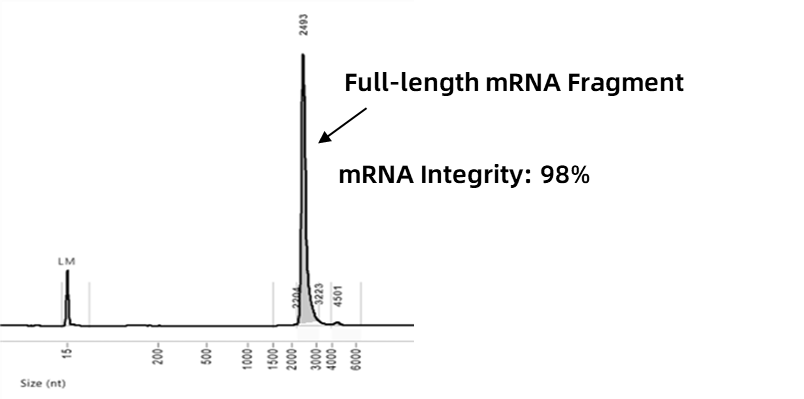
The integrity of mRNA directly influences its translation efficiency and protein function in cells. A structurally intact mRNA avoids abnormal folding or the formation of immunogenic dsRNA, thereby improving expression safety and reducing unintended immune responses.
Enorna ensures mRNA integrity >95% through process control, making it closer to its natural structure and enabling efficient and safe expression in cells.
Our comprehensive analytical platform provides thorough and rigorous testing services for RNA drug substance (DS), ensuring every batch meets predefined quality standards. We focus on Critical Quality Attributes (CQAs), including concentration, mRNA integrity, residual dsRNA, and both process-relate
Identification: Sequencing (Sanger Sequencing)
Purity: Residual dsRNA (ELISA), Integrity (Capillary Electrophoresis, CE)
Potency: Capping efficiency (LC-MS)
Safety: Residual host cell DNA, Endotoxin, Sterility
Reach Out Today
Contact us