

As critical starting material for nucleic acid drug manufacturing, purity, stability, and quality of plasmid DNA directly impact the safety, efficacy, and process stability of final drug product. Leveraging our mature plasmid production processes and robust quality management system, Enorna develops flexible production strategies tailored to different application scenarios and development stages. We provide clients with full plasmid CDMO services, spanning from preclinical research to commercial-scale production.


E.Coli Cell Banking


Seeding


Fermentation


Alkaline Lysis


Release Test


UF/DF


Chromatography


UF/DF
High-Density Fermentation: Optimized for maximizing plasmid yield.
Chromatographic Purification: Effectively remove impurities, including residual host cell DNA, RNA, proteins, endotoxin, etc.
Animal-Derived Component-Free (ADCF): Ensuring product safety and regulatory.
High Poly(A) Retention: Specialized processes to maintain the stability and integrity of Poly(A) tail.
Superior Linearization: Achieving a plasmid linearization efficiency of >95%.
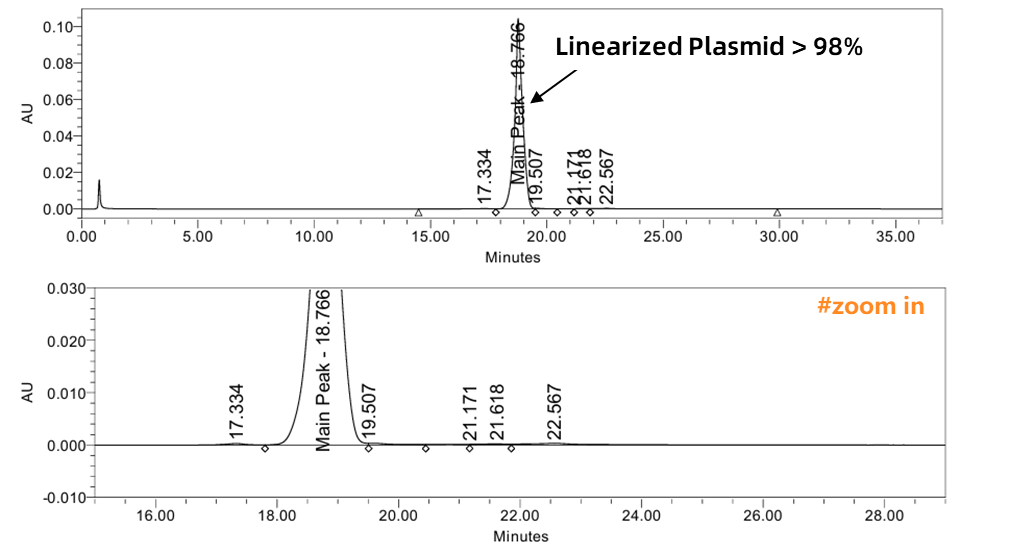
Plasmid linearization is a critical step in mRNA IVT. A higher linearization ratio leads to a more stable IVT process, superior mRNA product quality, lower immunogenicity, and reduced production costs.
By optimizing enzyme digestion systems and purification processes, Enorna maintains plasmid linearization ratios >95%, ensuring high-quality mRNA.
Phase-Appropriate Approach: Implementing flexible strategies to provide customized plasmid production solutions tailored to different application scenarios and development stages.
Scalable Supply: Flexible production scale ranging from milligram (mg) to gram (g) scales to meet diverse project requirements.
Reach Out Today
Contact us