

To improve the storage and transportation stability of nucleic acid drugs, Enorna has developed LNP lyophilization processes to overcome cold-chain limitations and enhance final product stability. Our fully closed aseptic fill-finish lines and automated lyophilization production platform provide one-stop services—from clinical and commercial filling to lyophilization—ensuring robust and stable drug product supply.
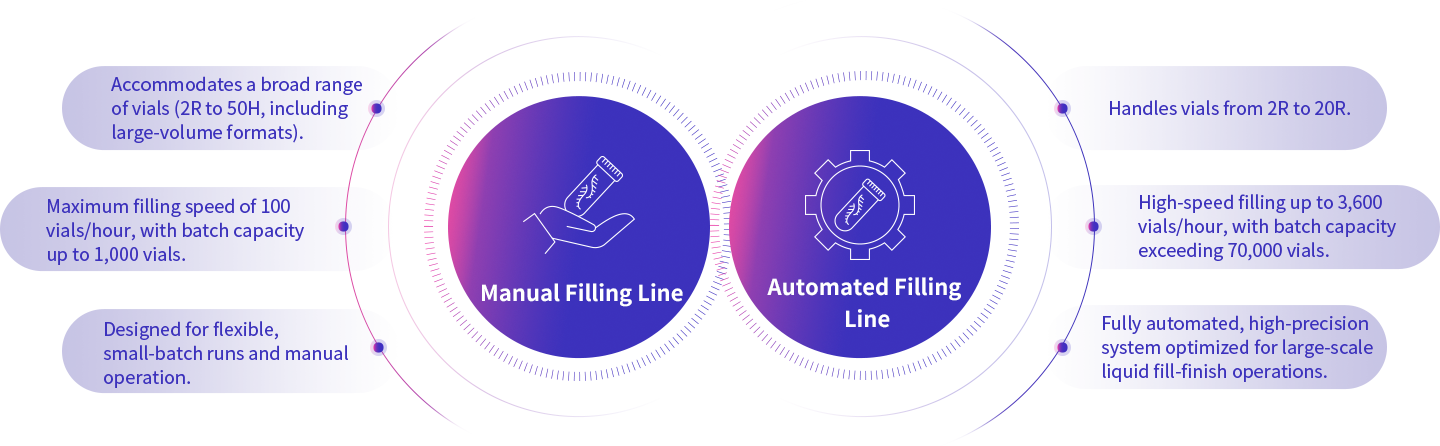
Enorna is equipped with both manual and automated filling lines, accommodating a wide range of container specifications and process requirements. Our capacity spans from small-scale production for Investigator-Initiated Trials (IIT, hundred-vial scale) to commercial-scale batches for market supply. We provide high-precision, high-consistency filling solutions that meet the most stringent requirements of cGMP and aseptic processing.
mRNA-LNP formulations have long relied on stringent low-temperature cold-chain conditions due to stability constraints. Enorna’s industry‑leading lyophilization technology overcomes this barrier by converting liquid LNPs into a stable solid dosage form. This eliminates ultra‑cold logistics, extends shelf‑life under refrigerated conditions, and improves global accessibility of nucleic acid therapies.


Maintains integrity at 2–8°C, enabling long‑term refrigerated storage.


Encapsulation efficiency and PDI remain stable through multiple freeze‑thaw cycles.


Pre‑defined lyophilization cycles minimize manual intervention, ensuring batch‑to‑batch consistency and cGMP compliance.


Integrated SIP/CIP systems sterilize the chamber and shelves per GMP standards, reducing cross-contamination risks significantly.


Integrated design of the lyophilizer and isolator filling system to prevent secondary contamination.